Dental infection treatment requires removing the infection source through drainage, root canal therapy, or extraction. Antibiotics serve only as adjunctive therapy for systemic involvement. Early intervention prevents serious complications like sepsis and tissue necrosis.
What Is a Dental Infection and Why Does It Matter?
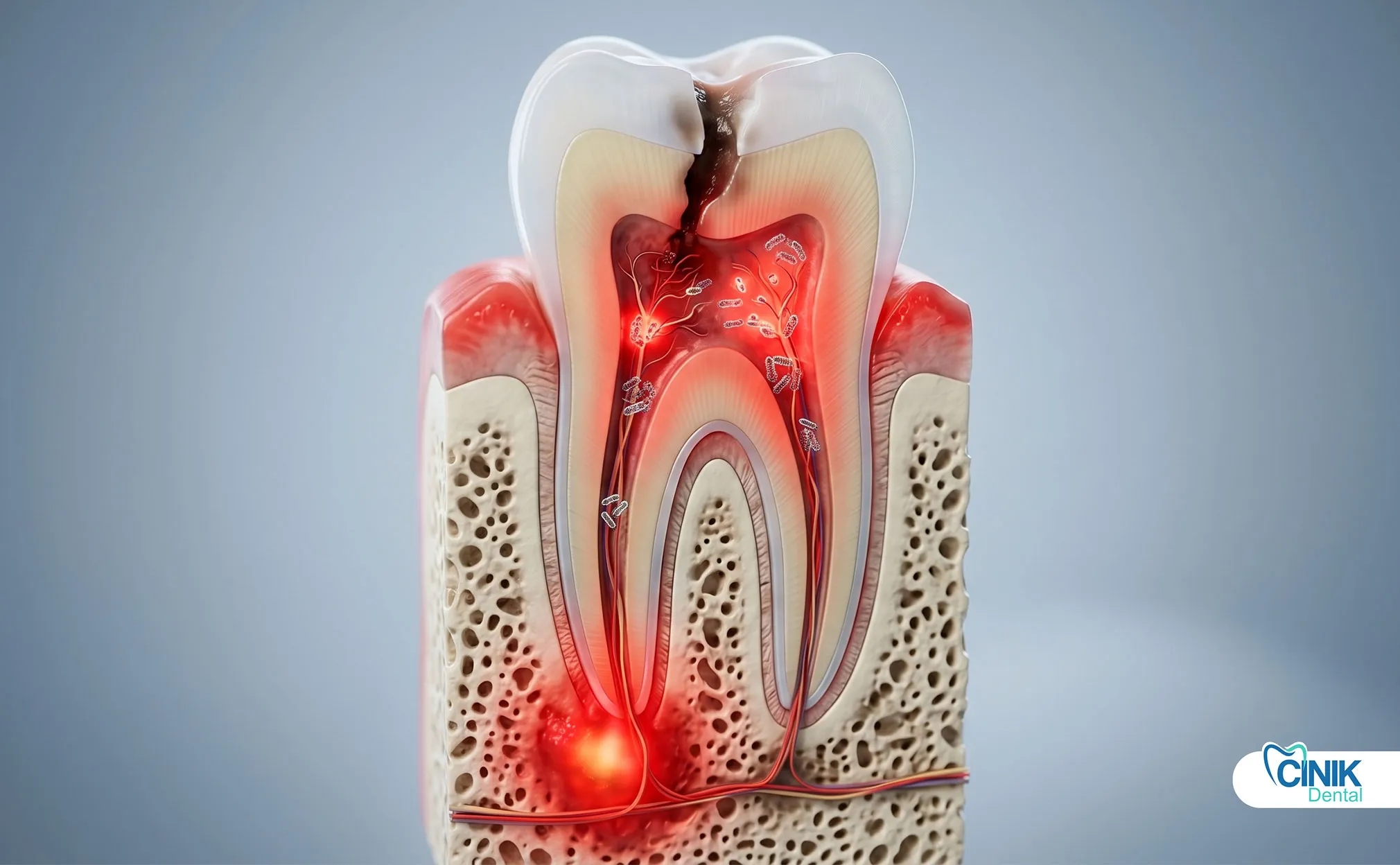
A dental infection is a bacterial invasion of tooth pulp, periodontal tissues, or surrounding bone. It causes severe pain, swelling, and can spread to facial spaces or bloodstream without prompt treatment.
A dental infection, clinically termed an odontogenic infection, develops when bacteria penetrate the protective layers of a tooth or surrounding gum tissue. These infections start small but can escalate into life-threatening conditions within days. The World Health Organization recognizes dental infections as a significant global health burden, particularly in populations with limited access to dental care (Petersen, 2022).
Dental infections fall into three primary categories based on location and origin. Periapical abscesses form at the root tip when pulp tissue dies from deep decay or trauma. Periodontal abscesses develop in the supporting structures of a tooth affected by advanced gum disease. Gingival abscesses remain confined to the gum margin, usually caused by foreign body impaction.
The clinical relevance of dental infections extends beyond oral discomfort. Research published in the Journal of Dental Research demonstrates that untreated dental infections increase cardiovascular risk, complicate diabetes management, and elevate systemic inflammatory markers (Lockhart, 2019). In 2023, emergency departments across the United States treated over 2 million cases of dental infections, with average treatment costs exceeding $1,000 per visit when hospitalization became necessary.
Early diagnosis matters because dental infections follow a predictable progression. Bacteria multiply in the confined space of a tooth root or gum pocket. Pressure builds. Tissue swells. The immune response triggers fever and malaise. Without intervention, the infection breaches local barriers and enters fascial spaces, lymphatic channels, or the bloodstream.
The treatment spectrum spans local interventions and systemic therapies. Local approaches, drainage, root canal therapy, extraction, eliminate the infection source directly. Systemic approaches, primarily antibiotics, support the body's defense when infection spreads beyond the tooth. The key principle: local treatment takes priority. Antibiotics alone cannot sterilize an infected root canal or abscess cavity.
Infection Type | Location | Primary Cause | Typical Treatment |
Periapical abscess | Root apex | Pulp necrosis | Root canal or extraction |
Periodontal abscess | Supporting structures | Advanced periodontitis | Drainage + periodontal therapy |
Gingival abscess | Gum margin | Foreign body impaction | Drainage + removal of irritant |
What Causes Dental Infections to Develop?
Dental caries, trauma, failed restorations, and periodontal disease allow bacteria to invade tooth pulp and surrounding tissues. Biofilm formation protects bacteria and accelerates tissue destruction.
How Do Bacteria Invade Dental Tissues?
Bacteria enter through enamel cracks, deep cavities, or gum pockets. They form biofilms that resist immune attacks and antibiotics, then destroy pulp, bone, and soft tissue.
Dental infections begin with bacterial colonization. The oral cavity hosts over 700 bacterial species, but only a handful cause invasive infections. Streptococcus mutans initiates tooth decay by metabolizing sugars into acid. Aggregatibacter actinomycetemcomitans and Porphyromonas gingivalis drive periodontal destruction. When these organisms breach enamel or gum defenses, they access the tooth pulp, a sterile chamber containing nerves and blood vessels.
Dental caries remains the leading cause of pulp infection. Untreated cavities progress through enamel, dentin, and finally into the pulp chamber. The pulp responds with inflammation, but bacterial toxins and pressure eventually kill the tissue. Necrotic pulp becomes a breeding ground for anaerobic bacteria.
Trauma accelerates infection risk. Cracked teeth, even without visible fractures, allow bacterial ingress. Failed restorations create microscopic gaps where bacteria accumulate. Root canal treatments that miss canal branches leave residual infection.
Periodontal disease provides an alternative pathway. As gum pockets deepen, bacteria migrate down the root surface. The immune system attacks both bacteria and host tissue, destroying the periodontal ligament and alveolar bone. Deep pockets exceeding 7 millimeters frequently harbor abscess-forming bacteria.
Biofilm formation complicates treatment. Bacteria secrete extracellular polysaccharides that form a protective matrix. This biofilm shields organisms from antibiotics, saliva antibodies, and immune cells. Within a biofilm, bacteria communicate through quorum sensing and share resistance genes. Research by Seneviratne (2017) in Periodontology 2000 confirms that biofilm-embedded bacteria demonstrate up to 1,000-fold greater antibiotic resistance than planktonic cells.
What Are the Main Types of Dental Infections?
Periapical abscesses affect root tips, periodontal abscesses attack supporting structures, and gingival abscesses stay at gum margins. Each type requires different treatment timing and approach.
Periapical Abscess: This infection forms at the apex of a tooth root after pulp necrosis. Pus accumulates in the bone surrounding the root tip. Patients experience throbbing pain that worsens when lying down. The tooth feels elevated in its socket. Swelling may appear on the facial skin if the infection perforates the bone. X-rays show a radiolucent area at the root apex. Root canal therapy or extraction resolves the infection source.
Periodontal Abscess: This infection develops in a pre-existing periodontal pocket. It causes rapid onset of pain, swelling, and purulent discharge from the gum margin. The affected tooth often has mobility due to bone loss. Periodontal abscesses require drainage through the gum pocket or an external incision. Scaling and root planing follow to reduce pocket depth.
Gingival Abscess: The most superficial type, confined to the gum margin. Foreign bodies, food impaction, toothbrush bristles, dental floss fragments, trigger localized infection. Pain remains moderate. Swelling stays limited. Treatment involves removing the foreign object and allowing natural drainage.
How Does a Dental Infection Spread?
Infections spread through local tissue planes, fascial spaces, and eventually the bloodstream. Facial swelling, difficulty swallowing, and fever signal dangerous progression.
Local tissue invasion represents the first phase. Bacteria and inflammatory mediators destroy surrounding bone and soft tissue. The abscess cavity enlarges. Pressure compresses nerves, causing intense pain. Enzymes like collagenase break down connective tissue barriers.
Fascial space involvement marks dangerous escalation. The face and neck contain multiple connective tissue compartments separated by fascial layers. Bacteria track along these planes, creating life-threatening swellings. The submandibular space infection (Ludwig's angina) can obstruct the airway. The cavernous sinus, accessed through facial vein drainage, risks thrombosis and brain infection.
Systemic dissemination occurs when bacteria enter blood vessels. Bacteremia, the presence of bacteria in blood, happens during dental procedures and even vigorous tooth brushing. In healthy individuals, immune cells clear transient bacteremia. In immunocompromised patients or those with severe infections, bacteremia progresses to sepsis. Endocarditis, infection of heart valves, poses particular risk for patients with prosthetic valves or previous cardiac damage.
Spread Stage | Location | Warning Signs | Risk Level |
Localized | Tooth and immediate bone | Tooth pain, mild swelling | Low |
Regional | Fascial spaces of face/neck | Facial swelling, trismus, dysphagia | Moderate-High |
Systemic | Bloodstream and distant organs | Fever, tachycardia, hypotension, confusion | Critical |
How Do Dentists Recognize and Diagnose Dental Infections?
Dentists use clinical examination, patient symptoms, radiographic imaging, and pulp testing to identify infection type, severity, and extent.
What Symptoms Indicate a Dental Infection?
Severe throbbing toothache, facial swelling, gum redness, temperature sensitivity, and fever indicate active infection requiring immediate evaluation.
Pain serves as the primary symptom. Infected teeth produce deep, throbbing pain that patients describe as relentless. The pain intensifies with heat exposure and may briefly decrease with cold application in early pulpitis. Once the pulp necrotizes, cold sensitivity disappears but pressure pain increases as pus builds under the root.
Swelling signals advancing infection. Early infections cause intraoral gum swelling. As pressure increases, swelling extends to facial skin. The skin becomes red, warm, and tense. Patients may report that swelling appeared suddenly and expanded over hours.
Systemic symptoms indicate spreading infection. Fever above 38°C (100.4°F) suggests immune mobilization against significant bacterial load. Malaise, loss of appetite, and difficulty opening the mouth (trismus) accompany fascial space involvement. Difficulty swallowing (dysphagia) or breathing represents emergency airway risk.
What Diagnostic Tools Confirm Dental Infections?
Clinical palpation, percussion testing, X-rays, CBCT scans, and electric pulp tests confirm infection presence, location, and tooth vitality status.
Clinical examination provides immediate clues. Dentists palpate swollen areas to assess fluctuance, soft, fluid-filled swelling indicates pus accumulation. Percussion testing taps teeth with a blunt instrument; infected teeth transmit vibration painfully through inflamed periodontal ligaments. Mobility testing identifies teeth with advanced bone loss from periodontal abscesses.
Radiographic imaging visualizes bone destruction. Standard periapical X-rays show radiolucent areas at root tips indicating bone loss from periapical abscesses. Bitewing radiographs reveal deep decay and periodontal bone levels. However, 2D X-rays miss early infections and complex anatomy.
Cone beam computed tomography (CBCT) offers three-dimensional visualization. This technology detects abscesses before visible bone changes appear on standard X-rays. CBCT identifies additional root canals, root fractures, and precise spread patterns into fascial spaces. Research by Patel (2019) in the International Endodontic Journal confirms CBCT detects 40% more periapical lesions than conventional radiography.
Pulp vitality testing distinguishes reversible from irreversible conditions. Electric pulp testers stimulate nerve response; non-vital teeth show no sensation. Cold testing with refrigerant spray assesses nerve function. A necrotic pulp fails to respond to cold, while an inflamed vital pulp produces exaggerated, lingering pain.
What Are the Core Principles of Dental Infection Treatment?
Remove the infection source first through drainage, root canal, or extraction. Use antibiotics only when infection spreads systemically. Never rely on antibiotics alone.
The primary goal of dental infection treatment is source control. Antibiotics circulate through blood but cannot penetrate necrotic pulp chambers, abscess cavities, or biofilm-protected bacterial colonies. The infection source, a dead pulp, accumulated pus, or diseased periodontal pocket, must be physically eliminated.
Drainage and debridement form the foundation of source control. An abscess cavity contains pus, bacterial toxins, and inflammatory fluid. Incision and drainage release this material, reducing pressure and bacterial load. Root canal therapy removes infected pulp tissue from within the tooth. Extraction eliminates the entire infected structure.
Conservative approaches preserve natural teeth when possible. Root canal therapy saves teeth with periapical abscesses if sufficient tooth structure remains for restoration. Periodontal abscesses often respond to drainage combined with deep cleaning. Extraction becomes necessary when teeth have fractures below the gum line, severe bone loss, or failed previous root canal treatment.
Surgical approaches address complex cases. Apicoectomy, surgical removal of the root tip, saves teeth with persistent periapical infection after root canal therapy. Periodontal surgery reduces deep pockets that harbor recurrent abscesses. Hospital-based surgery manages fascial space infections requiring airway protection.
Key Clinical Insight: Local treatment takes precedence over antibiotics. A patient with a dental abscess who receives only antibiotics experiences temporary symptom relief but faces guaranteed recurrence. The abscess cavity remains. Bacteria repopulate. Without drainage or tooth treatment, the infection returns, often with antibiotic-resistant organisms.
Which Dental Procedures Treat Infections First?
Dentists prioritize incision and drainage for abscesses, root canal therapy for savable teeth, and extraction for non-restorable teeth. These procedures eliminate infection sources directly.
How Does Abscess Drainage Work?
The dentist numbs the area, makes a small incision into the abscess, and allows pus to drain. This immediately reduces pain and pressure while removing bacterial load.
Incision and drainage provides the fastest pain relief for acute abscesses. The procedure requires local anesthesia. The dentist identifies the most fluctuant (softest, most fluid-filled) area of swelling. A scalpel incision, typically 1-2 centimeters, penetrates to the abscess cavity. Pus evacuates under pressure. The dentist may insert a small rubber drain to maintain opening for 24-48 hours.
Immediate benefits include pressure reduction and pain relief. Patients often report dramatic improvement within hours. Drainage also removes bacterial inoculum, reducing systemic bacterial load. The procedure takes 15-30 minutes in a dental office setting.
Post-drainage care involves warm saline rinses to maintain drain patency. Patients continue antibiotics if systemic symptoms exist. Definitive tooth treatment, root canal or extraction, follows once acute swelling subsides, usually within 3-7 days.
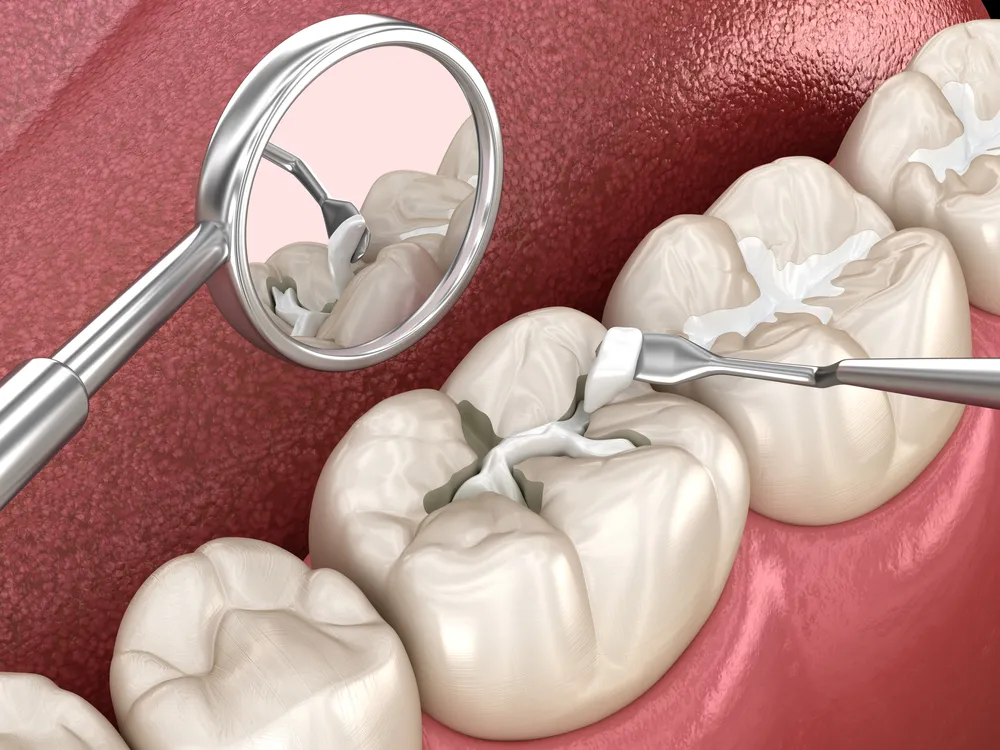
When Does Root Canal Therapy Save an Infected Tooth?
Root canal therapy removes infected pulp, disinfects root canals, and seals the tooth. It preserves natural teeth when sufficient structure remains for crown restoration.
Root canal therapy addresses periapical abscesses by removing the infected pulp chamber contents. The dentist creates an access opening through the tooth crown. Instruments remove pulp tissue, necrotic debris, and bacterial biofilm from the canal system. Chemical irrigants, sodium hypochlorite and chlorhexidine, disinfect canal walls.
The procedure requires 1-3 appointments depending on infection severity. First visit focuses on pulp removal and abscess drainage through the tooth. The dentist places antibacterial medication inside the canal and seals a temporary filling. Second visit confirms infection control before filling canals with gutta-percha, a rubber-like material. A permanent crown restores tooth strength.
Success rates exceed 90% for primary root canal treatments. Research by Ng (2011) in the International Endodontic Journal reports 94% survival at 4 years for teeth with periapical abscesses treated with root canal therapy and crown restoration. Failure typically stems from missed canal branches, persistent bacterial biofilm, or coronal leakage.
When Must an Infected Tooth Be Extracted?
Extraction becomes necessary when teeth have fractures below the gum line, severe bone loss, failed root canal retreatment, or insufficient structure for restoration.
Tooth extraction provides definitive infection elimination when preservation is impossible. Vertical root fractures, which extend below the gum line, prevent bacterial seal regardless of treatment. Severe periodontal disease destroying more than 50% of supporting bone makes teeth non-restorable. Failed root canal retreatment, particularly with persistent periapical lesions, may warrant extraction and implant replacement.
The extraction procedure for infected teeth requires care to prevent bacterial spread. The dentist may prescribe preoperative antibiotics for immunocompromised patients or severe infections. Local anesthesia with vasoconstrictor controls bleeding. The dentist elevates the tooth from its socket, removes granulation tissue from the socket base, and ensures complete debridement.
Post-extraction healing takes 1-2 weeks for soft tissue closure. Bone remodeling continues for 3-6 months. Immediate implant placement, performed in select cases, reduces treatment time but requires infection-free extraction sites.
Evidence-Based Note: Definitive dental procedures remain essential because antibiotics cannot eliminate infection sources. A systematic review by Cope (2014) in the British Dental Journal analyzed 22 clinical trials and concluded that antibiotics without source control provide no sustained benefit for dental abscesses. Symptom recurrence occurs in 100% of cases within weeks.
Procedure | Indication | Success Rate | Recovery Time |
Incision and drainage | Acute abscess with fluctuant swelling | Immediate relief; requires follow-up | 2-3 days |
Root canal therapy | Periapical abscess, restorable tooth | 90-95% at 5 years | 1-2 weeks |
Tooth extraction | Non-restorable tooth, failed retreatment | 100% infection elimination | 1-2 weeks soft tissue |
How Do Antibiotics Support Dental Infection Treatment?
Antibiotics treat spreading infections with fever, facial cellulitis, or immunocompromise. They never replace drainage or tooth treatment. Amoxicillin remains the first-line choice.
When Do Dentists Prescribe Antibiotics?
Dentists prescribe antibiotics for systemic symptoms, spreading cellulitis, immunocompromised patients, or when immediate dental procedures are impossible.
Antibiotics serve an adjunctive role in dental infection management. They do not cure dental abscesses. They control bacterial spread when the infection exceeds local containment. Specific indications include:
Systemic involvement: Fever above 38°C, malaise, or lymphadenopathy indicates bacterial load exceeding local immune capacity. Antibiotics reduce bacteremia risk and support systemic recovery.
Spreading infection: Cellulitis, diffuse skin redness without localized abscess, requires antibiotics because drainage is not possible. Infections tracking into fascial spaces need aggressive antibiotic coverage while awaiting surgical intervention.
Immunocompromised states: Patients with diabetes, HIV, chemotherapy, or organ transplants face higher sepsis risk. Early antibiotic intervention protects these vulnerable populations.
Procedural prophylaxis: Certain medical conditions require antibiotic coverage before dental procedures to prevent distant site infection. Prosthetic heart valves, previous infective endocarditis, and some orthopedic implants fall into this category.
Important Principle: Antibiotics are adjunctive, not primary therapy. A patient with a localized dental abscess and no systemic symptoms does not need antibiotics. The dentist performs drainage and tooth treatment. Adding antibiotics provides no benefit and contributes to resistance.
Which Antibiotics Work Best for Dental Infections?
Amoxicillin remains the first-line antibiotic due to effectiveness against typical oral pathogens and excellent tolerability. Penicillin V serves as an alternative.
Amoxicillin dominates first-line dental antibiotic prescribing. This aminopenicillin covers the gram-positive cocci (Streptococcus species) and gram-negative anaerobes (Prevotella, Fusobacterium) that cause most dental infections. Oral absorption reaches 90%, producing reliable blood levels. The standard adult dose is 500 mg three times daily for 5-7 days.
Clinical Evidence: A randomized controlled trial by Matthews (2016) in the Journal of Antimicrobial Chemotherapy compared amoxicillin with placebo for dental abscesses. Both groups received incision and drainage. Amoxicillin reduced post-procedure swelling by 35% and shortened recovery by 2 days. However, cure rates at 30 days were identical, confirming that source control, not antibiotics, determines outcomes.
Penicillin V provides an alternative with a narrower spectrum. This phenoxymethylpenicillin resists gastric acid degradation, making it suitable for oral use. The typical dose is 500 mg four times daily. Penicillin V covers most odontogenic pathogens but lacks gram-negative anaerobe coverage compared to amoxicillin.
What Antibiotics Help When First-Line Drugs Fail?
Metronidazole targets anaerobes and combines with amoxicillin for severe infections. Clindamycin replaces penicillin in allergic patients. Azithromycin offers another alternative.
Metronidazole specifically targets obligate anaerobic bacteria. These organisms dominate necrotic pulp and deep periodontal pockets. Metronidazole alone does not cover aerobic streptococci, so dentists often combine it with amoxicillin for severe, mixed infections. The dose is 400 mg three times daily. Patients must avoid alcohol during treatment and for 48 hours after completion due to disulfiram-like reactions.
Clindamycin serves penicillin-allergic patients. This lincosamide antibiotic covers gram-positive organisms and anaerobes effectively. The dose is 300 mg four times daily. Dentists reserve clindamycin for true penicillin allergies because it carries a higher risk of Clostridioides difficile colitis, a serious intestinal infection.
Azithromycin provides another alternative for penicillin-allergic patients. This macrolide antibiotic offers convenient dosing, 500 mg daily for 3 days, due to its long tissue half-life. However, rising resistance among oral streptococci limits its reliability. Dentists use azithromycin when clindamycin is contraindicated.
Why Does Antibiotic Resistance Threaten Dental Treatment?
Overprescription of dental antibiotics accelerates bacterial resistance. Guidelines now discourage routine antibiotic use for localized infections without systemic spread.
Antibiotic resistance undermines dental infection management. The Centers for Disease Control and Prevention identifies dental prescribing as a significant contributor to unnecessary antibiotic use. Studies estimate that 30-50% of dental antibiotic prescriptions are unnecessary, given for conditions where source control alone suffices.
The consequences extend beyond individual patients. Resistant bacteria colonize the oral cavity and spread to family members, healthcare facilities, and communities. Streptococcus pneumoniae and Staphylococcus aureus strains resistant to penicillin and clindamycin increasingly appear in dental infections.
Antibiotic stewardship programs target dental overprescription. The American Dental Association updated guidelines in 2019, explicitly stating that antibiotics are not indicated for localized intraoral swellings without systemic involvement. The guidelines emphasize: "The primary method for treating a dental infection should be source control."
Targeted therapy replaces empirical prescribing in recurrent or complex cases. Culture and sensitivity testing identifies specific bacteria and their resistance patterns. This approach, standard in hospital settings, is becoming more common in dental practice for immunocompromised patients or treatment failures.
Antibiotic | Spectrum | Typical Dose | Best For |
Amoxicillin | Broad (aerobes + anaerobes) | 500 mg TID x 5-7 days | First-line, most infections |
Penicillin V | Narrow (aerobes) | 500 mg QID x 5-7 days | Mild infections, penicillin preference |
Metronidazole | Anaerobes only | 400 mg TID x 5-7 days | Combined with amoxicillin for severe cases |
Clindamycin | Aerobes + anaerobes | 300 mg QID x 5-7 days | Penicillin allergy |
Azithromycin | Broad | 500 mg daily x 3 days | Penicillin allergy, clindamycin contraindicated |
How Do Dentists Manage Pain During Infection Treatment?
NSAIDs like ibuprofen provide superior dental pain relief compared to opioids. Acetaminophen offers an alternative for patients who cannot take NSAIDs. Local anesthesia during procedures eliminates procedural pain.
Pain management in dental infections follows a multimodal approach. Non-steroidal anti-inflammatory drugs (NSAIDs) form the cornerstone. Ibuprofen, 400-600 mg every 6-8 hours, reduces inflammation at the infection site while providing analgesia. Research by Moore (2015) in the Cochrane Database of Systematic Reviews confirms that ibuprofen 400 mg provides better pain relief than codeine 60 mg for dental pain, with fewer side effects.
Acetaminophen (paracetamol) serves patients with NSAID contraindications, peptic ulcer disease, anticoagulant therapy, or kidney impairment. The dose is 500-1000 mg every 6 hours, not exceeding 3000 mg daily. Acetaminophen lacks anti-inflammatory action but provides effective analgesia.
Combination therapy enhances pain control. Alternating ibuprofen and acetaminophen maintains steady drug levels. Some studies suggest concurrent administration provides superior relief to either drug alone, though patients must carefully track dosing intervals to avoid overdose.
Opioid prescribing has declined dramatically in dental practice. The opioid epidemic prompted reevaluation of dental pain protocols. Current guidelines reserve opioids for severe post-surgical pain unresponsive to NSAIDs. When prescribed, dentists limit quantities to 3-5 days and combine with NSAIDs to reduce opioid requirements.
Supportive care measures complement medication. Hydration maintains saliva flow and supports immune function. Warm saline rinses (half teaspoon salt in warm water, swished for 30 seconds) reduce bacterial load and promote drainage from gum pockets. Soft foods prevent mechanical irritation of infected tissues. Elevation of the head during sleep reduces blood flow to swollen facial tissues, decreasing throbbing pain.
What Happens When Dental Infections Go Untreated?
Untreated dental infections cause local bone destruction, tissue necrosis, and potentially fatal systemic complications including sepsis, airway obstruction, and brain abscess.
What Local Damage Do Untreated Infections Cause?
Untreated infections destroy jawbone (osteomyelitis), kill soft tissue (necrosis), and spread through facial spaces causing disfigurement and functional loss.
Osteomyelitis represents bone infection from bacterial seeding. The jawbone develops areas of dead bone (sequestra) surrounded by inflamed tissue. Chronic osteomyelitis causes persistent pain, draining fistulas, and pathologic fractures. Treatment requires prolonged antibiotics (4-6 weeks) and surgical removal of dead bone. Hyperbaric oxygen therapy supports healing in refractory cases.
Tissue necrosis occurs when infection compromises blood supply. The palate, tongue, or facial skin may develop black, devitalized areas requiring surgical debridement. Necrotizing fasciitis, though rare in dental infections, destroys tissue planes rapidly and demands emergency surgical intervention.
Facial space infections distort anatomy. The submandibular, pterygomandibular, and canine spaces swell dramatically. Patients develop "bull neck" appearance from bilateral submandibular involvement. Trismus, jaw locking from muscle inflammation, prevents eating and speaking.
What Systemic Risks Do Dental Infections Create?
Bacteria from dental infections enter the bloodstream, causing sepsis, heart valve infection, and distant abscesses. Airway obstruction from neck swelling can cause suffocation.
Sepsis represents the most feared systemic complication. Bacterial toxins trigger massive immune activation. Fever, rapid heart rate, rapid breathing, and confusion signal sepsis. Blood pressure drops. Organs fail. Septic shock carries 40% mortality even with intensive care treatment. Dental infections cause 10-15% of all facial sepsis cases.
Airway obstruction threatens life within hours. Ludwig's angina, a bilateral submandibular space infection, pushes the tongue upward and backward. The floor of the mouth becomes board-like. Patients drool, cannot swallow saliva, and develop stridor (noisy breathing). Emergency tracheostomy or intubation secures the airway before surgical drainage.
Brain abscess, though rare, follows hematogenous spread or direct extension through skull bones. Frontal tooth infections access the frontal lobe. Maxillary infections reach the temporal lobe. Brain abscesses cause headache, seizures, focal weakness, and altered mental status. Neurosurgical drainage and prolonged antibiotics (6-8 weeks) treat this complication.
Endocarditis affects heart valves, particularly in patients with pre-existing valve disease or prosthetic valves. Viridans streptococci from dental infections adhere to damaged endocardium, forming vegetations that embolize to brain, kidneys, and limbs. Prophylactic antibiotics before dental procedures prevent this complication in high-risk patients.
Complication | Mechanism | Mortality Risk | Treatment Required |
Osteomyelitis | Bone infection and necrosis | Low | Surgery + 4-6 weeks antibiotics |
Sepsis | Bloodstream bacterial spread | 15-40% | ICU care, IV antibiotics, source control |
Ludwig's angina | Bilateral submandibular infection | 10-20% | Emergency airway + surgical drainage |
Brain abscess | Hematogenous or direct spread | 10-30% | Neurosurgery + 6-8 weeks antibiotics |
How Should Special Populations Receive Dental Infection Care?
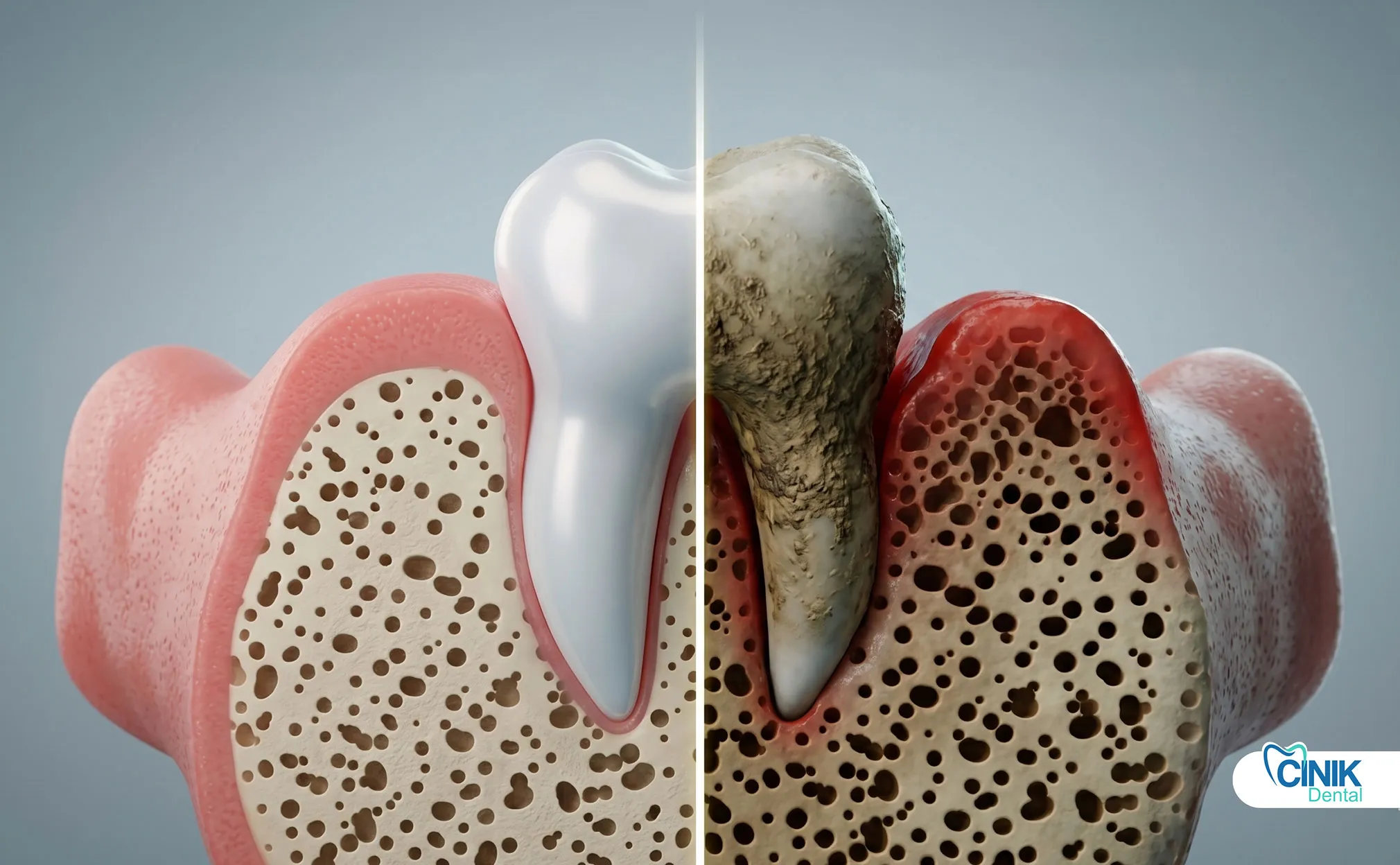
Immunocompromised, pediatric, and elderly patients need modified treatment approaches, earlier antibiotic intervention, adjusted drug dosing, and closer monitoring.
What Extra Care Do Immunocompromised Patients Need?
Immunocompromised patients face higher systemic spread risk. They need earlier antibiotic starts, broader coverage, hospital-based treatment for moderate infections, and longer recovery monitoring.
Immunocompromised states include diabetes mellitus, HIV/AIDS, chemotherapy, organ transplantation, chronic corticosteroid use, and inherited immune deficiencies. These conditions impair neutrophil function, antibody production, or cell-mediated immunity, critical defenses against dental pathogens.
Higher infection spread risk demands aggressive management. A localized abscess in a healthy patient may remain confined for days. The same abscess in a neutropenic chemotherapy patient can progress to sepsis within hours. Dentists lower thresholds for antibiotic initiation and hospital referral.
Early antibiotic intervention uses broader spectrum coverage. While healthy patients receive amoxicillin alone, immunocompromised patients may need amoxicillin-clavulanate (Augmentin) for enhanced anaerobe coverage, or even intravenous antibiotics for hospital admission.
Hospital-based treatment becomes necessary for infections that would be office-managed in healthy patients. IV access provides reliable drug delivery. Monitoring tracks temperature, white blood cell counts, and inflammatory markers. Surgical teams stand ready for airway management if fascial space involvement develops.
How Do Dentists Adjust Treatment for Children and Elderly Patients?
Children require weight-based dosing and behavior management. Elderly patients need renal function assessment, drug interaction review, and consideration of comorbidities.
Pediatric dental infections present unique challenges. Primary teeth with periapical abscesses often require extraction rather than root canal therapy due to complex root anatomy and the approaching exfoliation timeline. Space maintainers prevent orthodontic complications after premature tooth loss. Antibiotic dosing follows weight-based calculations, amoxicillin 50 mg/kg/day divided into 3 doses.
Behavior management enables treatment in young patients. Local anesthesia requires careful technique to avoid intravascular injection in small patients. Sedation or general anesthesia may be necessary for extensive procedures. Parents play crucial roles in post-treatment care, ensuring medication compliance and dietary modifications.
Geriatric patients present different complexities. Polypharmacy increases drug interaction risks. Antibiotics may interact with warfarin (clindamycin and metronidazole potentiate effects), statins, or cardiac medications. Renal function decline requires dose adjustments for renally-cleared drugs like amoxicillin and metronidazole.
Cognitive impairment complicates symptom reporting. Elderly patients with dementia may not communicate pain effectively. Caregivers must observe behavioral changes, refusing food, facial touching, or agitation, as infection indicators. Treatment planning prioritizes simplicity and durability over idealistic approaches.
Population | Key Consideration | Antibiotic Adjustment | Special Needs |
Immunocompromised | High spread risk | Broader spectrum, earlier start | Hospital referral threshold lowered |
Pediatric | Growth and development | Weight-based dosing | Behavior management, space maintenance |
Geriatric | Comorbidities, polypharmacy | Renal dose adjustment | Drug interaction review, cognitive assessment |
How Can People Prevent Dental Infections?
Daily brushing and flossing, regular dental check-ups every 6 months, prompt cavity treatment, and gum disease management prevent the conditions that lead to dental infections.
Prevention of dental infections targets the root causes: tooth decay and periodontal disease. Oral hygiene practices disrupt bacterial biofilm formation before it causes tissue damage. The American Dental Association recommends brushing twice daily with fluoride toothpaste for 2 minutes, using a soft-bristled brush in gentle circular motions at the gumline.
Flossing reaches areas brushes miss, between teeth and below gum margins where periodontal pathogens colonize. Water flossers and interdental brushes assist patients with limited manual dexterity. Mouth rinses containing chlorhexidine or essential oils reduce bacterial load but do not replace mechanical cleaning.
Regular dental check-ups enable early intervention. Professional cleanings remove calculus (tartar) that harbors bacteria. Examinations detect cavities before they reach the pulp. Bitewing X-rays identify interproximal decay invisible to visual inspection. The 6-month recall interval suits most patients, though those with high decay risk or active periodontal disease may need 3-4 month intervals.
Early treatment of caries prevents pulp infection. Small cavities require simple fillings. Delay allows decay to deepen, eventually necessitating root canal therapy or extraction. Dental sealants on posterior teeth protect children and adolescents from occlusal surface decay.
Periodontal disease management prevents periodontal abscesses. Scaling and root planing (deep cleaning) removes calculus from root surfaces. Periodontal maintenance every 3 months controls disease progression. Surgical pocket reduction eliminates deep spaces where abscesses form.
Dietary modifications support prevention. Limiting sugar exposure, particularly frequent snacking and sugary beverages, reduces Streptococcus mutans acid production. Chewing sugar-free gum stimulates saliva flow, which neutralizes acid and remineralizes enamel. Fluoridated water strengthens tooth structure against acid attack.
What Is the Expected Recovery After Dental Infection Treatment?
Pain improves within 24-48 hours after source control. Soft tissue heals in 1-2 weeks. Bone healing takes 3-6 months. Early intervention produces faster, more complete recovery.
Expected healing timelines vary by procedure and infection severity. Drainage of an acute abscess produces immediate pain relief. Swelling subsides over 2-3 days. The small incision heals within a week. However, the underlying tooth still requires definitive treatment to prevent recurrence.
Root canal therapy recovery follows a predictable pattern. Post-operative soreness lasts 2-3 days, manageable with NSAIDs. The periapical bone begins healing within weeks, though complete radiographic bone fill requires 6-12 months. The final crown placement, 2-4 weeks after root canal completion, restores function and prevents fracture.
Extraction sites heal in stages. Blood clot formation occurs immediately. Soft tissue closes over 1-2 weeks. Bone fills the socket over 3-6 months. Implant placement, when planned, typically occurs 3-4 months post-extraction to allow initial bone healing.
Early intervention dramatically improves outcomes. A periapical abscess treated within days of symptom onset responds to simple root canal therapy. Delay of weeks allows bone destruction to expand, complicating treatment and reducing tooth survival probability. Periodontal abscesses treated promptly preserve teeth; delayed treatment often results in hopeless prognosis requiring extraction.
Factors affecting outcomes include patient compliance, immune status, and follow-through with definitive care. Patients who complete prescribed antibiotic courses and return for scheduled definitive procedures experience better results. Smoking impairs healing by reducing blood flow and immune function. Diabetes requires glycemic control for optimal recovery.
What Advances Are Shaping Future Dental Infection Treatment?
New antimicrobial peptides target biofilms without resistance. Laser-assisted disinfection improves root canal sterilization. Personalized medicine uses genetic testing to predict infection risk and treatment response.
Advances in antimicrobial therapy address biofilm resistance. Traditional antibiotics fail against biofilm-protected bacteria. Researchers develop antimicrobial peptides, short protein fragments that disrupt bacterial membranes without resistance development. These peptides, derived from natural immune molecules, show promise as root canal irrigants and topical treatments.
Photodynamic therapy uses light-activated dyes to kill bacteria. The dentist applies a photosensitizing agent to the infected area, then exposes it to specific light wavelengths. Reactive oxygen species destroy bacterial membranes and DNA. This technique sterilizes root canal systems and periodontal pockets without antibiotics.
Minimally invasive techniques preserve tooth structure. GentleWave technology uses multisonic energy and advanced fluid dynamics to disinfect complex root canal anatomy with minimal instrumentation. Laser-assisted root canal treatment vaporizes infected tissue and sterilizes canal walls simultaneously.
Personalized dental care leverages genetic and microbiome analysis. Genetic testing identifies patients with innate immune deficiencies predisposing to severe infections. Oral microbiome profiling predicts periodontal disease risk and guides targeted probiotic therapy. Pharmacogenomics determines optimal antibiotic selection based on individual metabolism.
Regenerative endodontics represents a paradigm shift. Instead of removing pulp and filling canals with inert material, this approach encourages pulp tissue regeneration using stem cells and growth factors. Immature teeth with infected pulps can continue root development, producing stronger, more durable teeth. Research by Galler (2021) in the Journal of Endodontics demonstrates continued root maturation in 80% of treated immature permanent teeth.
Frequently Asked Questions About Dental Infection Treatment
Do All Dental Infections Require Antibiotics?
No. Localized dental abscesses without fever or facial swelling need only source control, drainage, root canal, or extraction. Antibiotics help only when infection spreads systemically.
The misconception that every dental infection needs antibiotics drives overprescription. Clinical guidelines from the American Dental Association and the British National Institute for Health and Care Excellence explicitly state that antibiotics are unnecessary for most localized dental infections. Source control procedures eliminate the infection directly. Adding antibiotics provides no benefit and contributes to resistance.
Antibiotics become necessary when patients show systemic involvement. Fever, malaise, lymphadenopathy, or spreading cellulitis indicate bacterial load exceeding local containment. Immunocompromised patients may need earlier antibiotic starts. When dentists cannot perform immediate source control due to patient access issues, antibiotics serve as temporary bridges to definitive care.
How Long Does Dental Infection Treatment Take?
Acute abscess drainage provides immediate relief. Root canal therapy requires 1-3 appointments over 1-2 weeks. Extraction heals in 1-2 weeks. Antibiotic courses last 5-7 days.
Treatment duration depends on infection severity and chosen approach. Simple drainage of an abscess takes 30 minutes in the dental office. Pain improves within hours. However, the patient must return for definitive tooth treatment within days to prevent recurrence.
Root canal therapy typically spans two appointments. The first visit removes infected pulp and places antibacterial medication. The second visit, 1-2 weeks later, fills and seals the canals. A third appointment places the permanent crown 2-4 weeks later. Total treatment time: 4-8 weeks for complete restoration.
Extraction provides the fastest definitive solution. The procedure takes 20-40 minutes. Soft tissue heals in 1-2 weeks. If implant replacement is planned, the timeline extends 3-6 months for bone healing before implant placement.
Antibiotic courses remain short. Five to seven days suffices for dental infections, unlike the prolonged courses needed for deep tissue or bone infections. Patients must complete the full course even if symptoms improve earlier to prevent recurrence and resistance.
Can Dental Infections Heal on Their Own?
No. Dental infections require professional intervention. The body cannot eliminate infection sources inside teeth or abscess cavities. Delayed treatment increases complication risk.
Occasionally, an abscess ruptures spontaneously, draining pus and temporarily relieving pressure. Patients may interpret this as "healing." However, the infection source, necrotic pulp, deep decay, or periodontal pocket, remains. Bacteria repopulate. The abscess recurs, often with more resistant organisms.
The enclosed environment of a tooth root or abscess cavity prevents immune access. Neutrophils and antibodies cannot penetrate necrotic tissue or thick pus effectively. Antibiotics circulating in blood cannot reach bacteria in these avascular spaces. Only mechanical removal, drainage, root canal, or extraction, eliminates the infection.
Rarely, chronic low-grade infections persist for months without acute symptoms. These "silent" infections still destroy bone and release inflammatory mediators into the bloodstream. They contribute to systemic health problems without causing noticeable pain. Regular dental X-rays detect these hidden infections.
When Should Someone Seek Emergency Care for a Dental Infection?
Seek emergency care for difficulty breathing or swallowing, rapidly spreading facial swelling, fever above 39°C (102°F), confusion, or eye involvement from upper tooth infections.
Emergency warning signs indicate life-threatening progression. Airway compromise from submandibular or floor-of-mouth swelling requires immediate emergency department evaluation. Patients who cannot swallow their own saliva, who develop noisy breathing (stridor), or who feel their throat closing need emergency airway management.
Rapidly spreading cellulitis, swelling that visibly expands over hours, suggests aggressive bacterial strain or compromised immunity. IV antibiotics and possible surgical drainage become necessary. Facial swelling crossing the midline or involving both sides indicates bilateral spread.
Systemic toxicity signals sepsis. High fever, rapid heart rate (over 100 beats per minute), rapid breathing (over 20 breaths per minute), and altered mental status demand emergency care. These patients need blood cultures, IV fluids, and broad-spectrum antibiotics in an intensive care setting.
Eye involvement from upper tooth infections threatens vision. Swelling around the eye, double vision, or inability to move the eye indicates orbital cellulitis. This complication, arising from maxillary tooth infections spreading through thin bone into the orbit, requires emergency ophthalmology and ENT consultation.
Symptom | Action Required | Timeframe |
Toothache with mild swelling | Dental appointment | Within 24-48 hours |
Facial swelling, fever | Urgent dental or medical care | Same day |
Difficulty breathing/swallowing | Emergency department | Immediately |
Eye swelling, vision changes | Emergency department | Immediately |
Confusion, high fever, rapid heart rate | Emergency department/911 | Immediately |
Conclusion: Why Early Action Saves Teeth and Lives
Dental infections demand prompt source control through drainage, root canal, or extraction. Antibiotics play a supporting role for systemic spread. Prevention through regular care avoids the pain, cost, and danger of advanced infections.
Dental infection treatment rests on clear principles. Remove the infection source. Drain pus and necrotic tissue. Preserve natural teeth when possible. Extract when necessary. Use antibiotics judiciously, only for systemic involvement or when procedures must be delayed.
The evidence overwhelmingly supports this approach. Antibiotics alone fail because they cannot reach bacteria in necrotic pulp chambers and abscess cavities. Biofilm-protected organisms resist drug penetration. Only mechanical removal of infected tissue produces cure.
Early diagnosis prevents complications. A small periapical lesion on X-ray warns of impending abscess. A deep periodontal pocket signals abscess risk. Treating these conditions before acute symptoms develop saves teeth, money, and health.
Balanced antibiotic use protects individual patients and public health. Every unnecessary prescription contributes to resistance. Dentists who follow stewardship guidelines, prescribing only for indicated cases, choosing narrow-spectrum first-line drugs, and limiting duration, preserve antibiotic effectiveness for future generations.
Integration of clinical treatment and prevention offers the best outcomes. Patients who combine regular dental visits with daily home care rarely develop infections. When infections occur, they seek care promptly rather than enduring pain or hoping for spontaneous resolution.
The future of dental infection treatment looks promising. Biofilm-targeting therapies, regenerative techniques, and personalized medicine approaches will enhance outcomes while reducing antibiotic dependence. However, the fundamental principle remains unchanged: identify the source, remove the infection, and restore health.
References
Cope, Anwen L., et al. "Antibiotic Use in Dental Practice: A Systematic Review." British Dental Journal, vol. 216, no. 9, 2014, pp. 523-30.
Galler, Kerstin M., et al. "Regenerative Endodontic Procedures for the Treatment of Pulp Necrosis in Immature Permanent Teeth: A Systematic Review." Journal of Endodontics, vol. 47, no. 8, 2021, pp. 1193-202.
Lockhart, Peter B., et al. "Poor Oral Health as a Chronic, Potentially Infectious and Inflammatory Condition." Journal of Dental Research, vol. 98, no. 10, 2019, pp. 1073-80.
Matthews, David C., et al. "Antibiotics for Acute Dental Abscesses: A Systematic Review." Journal of Antimicrobial Chemotherapy, vol. 71, no. 3, 2016, pp. 564-71.
Moore, R. Andrew, et al. "Non-Steroidal Anti-Inflammatory Drugs for Acute Dental Pain." Cochrane Database of Systematic Reviews, no. 12, 2015, CD004978.
Ng, Yuan-Ling, et al. "Outcome of Primary Root Canal Treatment: Systematic Review of the Literature." International Endodontic Journal, vol. 44, no. 7, 2011, pp. 597-612.
Patel, Shanon, et al. "Cone Beam Computed Tomography in Endodontics." International Endodontic Journal, vol. 52, no. 9, 2019, pp. 1248-64.
Petersen, Poul Erik. "World Health Organization Global Policy for Improvement of Oral Health." International Dental Journal, vol. 72, no. 4, 2022, pp. 302-07.
Seneviratne, Chaminda J., et al. "Dental Plaque Biofilm: An Archetypal Biofilm." Periodontology 2000, vol. 75, no. 1, 2017, pp. 34-44.